The First Electric Glow Lamp
Many people alive today will vividly remember the thrill awakened by the first appearance of the electric glow lamp-the whole world felt the thrill. To see a little thread of carbon in a bulb become brilliantly incandescent when electric current passed through it was to taste in its fullness the mystery and the wonder of electricity. This triumph of invention, which had been sought for years by numerous workers, was shared by Great Britain and the United States. Almost simultaneously the late Sir Joseph Swan and America’s most popular inventor, T. A. Edison, succeeded in the quest. Others-notably Lane-Fox and Hiram Maxim-contributed to the achievement; but it was mainly under the patents of Swan and Edison that the electric lamp industry was first developed.
This was the period of monopoly, when other workers in the electric lamp field had either to cease operations or go abroad. Among those who took the latter course was the late Mr. C. J. Robertson, who had been associated with Mr. Lane-Fox. He went to the Continent, where he equipped and managed a number of factories and so gained experience which proved invaluable when, in 1893, the original carbon lamp patents expired.

First Generation Phosphors
The earliest fluorescent tubes employed a phosphor of Zinc Beryllium Silicate – the first material found to create white light with reasonable quality. However the efficacy of the popular 5-foot tube using these materials was just 35 lumens per watt. These phosphors were rapidly superseded once more efficient materials had been developed.
Second Generation – The Halophosphates
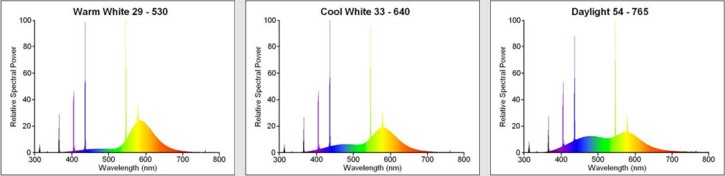
The most commonly found tubes on the global lighting market employ an internal coating of Calcium Halophosphate materials (generally known simply as ‘Halophosphate’ tubes). This revolutionary material was invented in 1942 by a group led by A.H. McKeag of Osram-GEC in London, and succeeded in almost doubling lamp efficacy. More than any other invention, it is this breakthrough which is responsible for having propelled the fluorescent business into the vast market it enjoys today. However by modern standards, the halophosphate materials are relatively inefficient and deliver inferior lighting quality compared to newer technologies. Although such tubes have a low initial purchase cost, this is rapidly offset by the increased electrical power consumption required to generate a given amount of light. Owing to their reduced energy efficiency, Halophosphate tubes will shortly be phased out under EU legislation to enforce the adoption of other more efficient fluorescent phosphor materials.

A wide variety of different whites can be created by the halophosphate system. The phosphor is in fact a blend of two different materials which radiate broadly in the blue and orange parts of the spectrum respectively. By changing the ratio of the two components a full range of warm to cool white hues can be achieved. Some typical spectral power distributions for the Warm, Cool and Daylight White Halophosphate lamps are illustrated in Figure Below, which reveals the changing ratio of the blue and orange radiating components. The colour rendering index is typically 50 to 70 and the lamp efficacy approx. 60 – 75 lm/W.
Third Generation – The Triphosphors
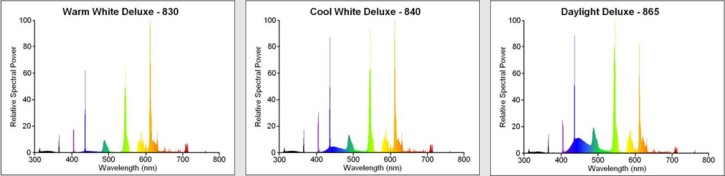
During the 1970s a great deal was learned about human vision systems, and this spurred fluorescent research into new territories. Traditionally it had been believed that to achieve good colour rendering, it was important for the lamp to produce a full spectrum with light of all wavelengths present. However the human eye is not equally sensitive to all colours. Therefore if a lamp is built to radiate all wavelengths, much of it will occur in regions to which the eye is less sensitive. New insights into the perception of colour vision discovered that it should be possible to achieve a very high colour rendering index from a light source having a narrow tri-band spectrum. These three bands correspond to the red, green and blue photo-receptors in the eye. It was theoretically proposed by the Westinghouse company in the USA to develop a prime-colour tube with three peaks at 450nm (blue), 540nm (green) and 610nm (orange) to coincide with the mechanisms of human colour vision.

New materials were synthesised with emissions close to these targets, and the modern family of Triphosphor lamps was born. One of each of the following blue, green and red fluorescent components are generally employed in modern triphosphor tubes:
| Colour | Phosphor Name | Chemical Formula | Wavelength |
| Blue | Barium Aluminate (BAM) | BaMg2Al16O27 : Eu2+ | 450nm |
| or | SrCaBaMg Chloroapatite | (Sr,Ca,Ba,Mg)5(PO4)3Cl : Eu2+ | 453nm |
| Green | Calcium Tungstate (CAT) | Ce0.65Tb0.35MgAl11O19 | 543nm |
| or | Lanthanum Phosphate (LAP) | LaPO4 : Ce3+Tb3+ | 544nm |
| Orange-Red | Yttrium Oxide (YOX) | Y2O3 : Eu3+ | 611nm |
By blending together the blue, green and red components in the correct proportions, a net white output of various hues can be realised. The distinctive triple-peak spectra of the triphosphor colours are illustrated in Figure Below for three popular shades of white. Note that the cooler the colour temperature, the greater the proportion of blue light in the spectrum.
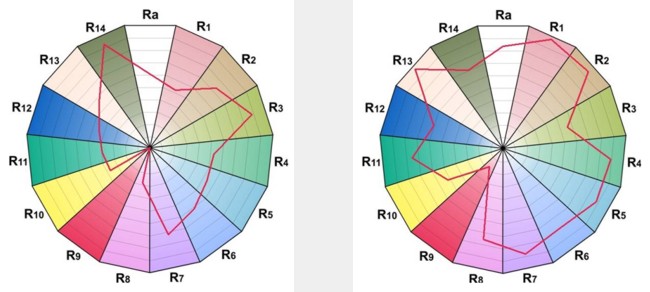
Owing to the proximity of these peaks to the colour receptors in the human eye, a very high colour rendering index is achieved. Typically this is of the order of Ra85 for most products, which marks a considerable improvement over Halophosphate materials. The two CRI wheel diagrams in Figure Below compare the colour rendering quality of a typical neutral white triphosphor tube with a halophosphate lamp delivering the same colour temperature. It will at once be appreciated that the triphosphor technology shows far superior rendition of almost all of the fourteen CIE test colours. In particular they offer an enormous improvement in the score for the R9 index, representing saturated red colours. Triphosphor lamps offer tremendous added value in applications where colour rendering quality is important, for instance a retail sales environment.
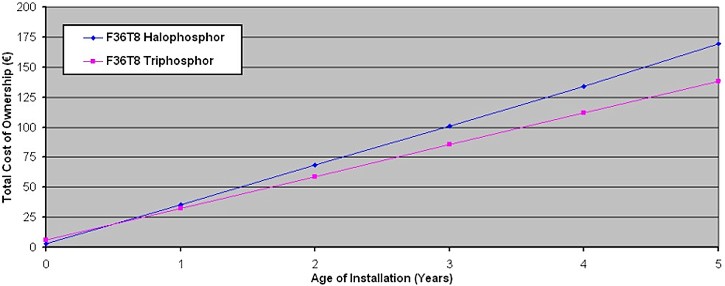
An unexpected advantage of these deeply saturated coloured phosphors was their astonishing efficiency of converting UV into visible light. As a result lamp efficacy is boosted to levels of around 80 – 95 lm/W. Perhaps the only reason for the lack of penetration of triphosphor materials is their high cost. It is often said that the cost of coating a fluorescent lamp with ordinary halophosphor mixtures is analgous with the price of flushing the glass tubes with milk – whereas the triphosphor solutions are more akin to filling the tubes with a good scotch whisky! This is due largely to the scarcity and cost of the rare earth activator materials employed in the triphosphor components, such as Europium, Cerium and Terbium. The pure cost difference of triphosphor lamps puts off many consumers who prefer to remain with basic products, but really this is a false economy. The less efficient halophosphate tubes consume far more than this initial investment through their greater electrical power consumption that is required to produce a given amount of light during lifetime. The rapid payback interval for triphosphor tubes is illustrated in the cost of ownership graphic in Figure Below. It can be seen that while the triphosphor lamps cost roughly twice as much to purchase, this initial expenditure is rapidly recouped through lower electricity consumption costs for a fixed lighting level.
Fourth Generation – Very High CCT
Current research into light and its effects on the human psyche are continuing to drive lamp technology into exciting new areas, and recent years have witnessed a number of special colour tubes which are intended to create an enhanced sense of well-being. Originally developed to treat the medical condition SAD (Seasonal Affective Disorder), they feature a very high colour temperature thanks to a boosted blue component. The actual colour temperature of these new tubes is pitched between 8000K and 17000K depending on the manufacturer.
The balance of phosphors has been engineered to strike what is currently believed to be the optimal balance between light for vision and light for personal well-being. Tests carried out in factories, offices and schools reveal an astonishing increase in personal performance of 10% or more, with the subjects feeling more alert, awake and energised.

These new tubes are beginning to create a positive influence especially in windowless offices and dull classrooms, where their enhanced blue component mimics the presence of these wavelengths in natural daylight. However as with all other colours, it is essential to bear in mind the teachings of the Kruithof diagram when installing light of such a high colour temperature. Simply retrofitting these tubes into conventional luminaires can produce negative visual effects, because the human eye requires much more light as the colour temperature is increased. Careful consideration must be given to the lux level on the working plane to ensure that compatibility is maintained with the very high colour temperature being provided.
In certain cases they can be mixed with warmer colour tubes with intelligent controls to achieve light levels and colours which match natural daylight conditions depending on the seasons and the time of day.
Coloured Light Sources
The flexibility of fluorescent technology lends itself well to the generation of light colours other than white. A full range of coloured tubes exists, as well as certain ultra-violet versions. These have diverse applications such as sun tanning, photocopying, medical treatments, detection of forged bank notes, identification of minerals, entertainment lighting etc. The spectra of some common tube colours are shown in the diagrams below, and their colour points are indicated on the CIE chromaticity triangle.
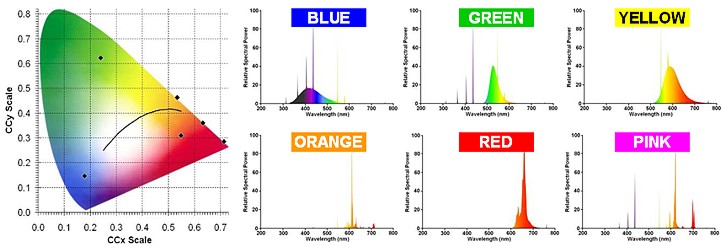
TRIPHOSPHOR LAMPS
The colour qualities of a lamp are characterised by two different aspects – colour temperature and colour rendering.
| Lamp Type | Approx. Colour Temperature | Colour Rendering (Ra) |
| Incandescent (240V) | 2800 degree K | 100 |
| Incandescent 12V | Dichroic 3000 to 4000 degree K | 100 |
| Warm White Fluorescent | 3300 degree K and below | 50 to 90 |
| Cool White Fluorescent | 3500 to 4000 degree K | 60 to 90 |
| Daylight Fluorescent (Cold White) | 6000 degree K and above | 80 to 90 |
The colour rendering of fluorescent lamps in the table above varies from different manufacturers.
Application of Different Colour Temperature Lamps
- Use warm white for accentuating warm colours (red, brown cream etc.)
- Use cool white with cool colours like grey and blue
- Use daylight colours in hot climates as a warm lamp makes people feel hotter
Colour Rendering
Fluorescent lamps do not emit equal intensities over the colour spectrum, but have peaks and valleys across the visible spectrum. This causes unnatural appearance (rendering) of coloured objects when viewed under fluorescent lamps.
Colour rendering is the ability of a lamp to give good colour representation of the object it is illuminating. The unit of measurement is Ra.
Some lamps have better colour rendering than others and should be selected for colour critical applications.
| Ra | Application |
| Above 90 | Colour Matching, Picture Galleries |
| 80 – 90 | Homes, Restaurants, Textile Industry |
| 60 – 80 | Offices, Schools, Light Industry |
| 40 – 60 | Heavy Industry |
| 20 – 40 | Outdoor |
International Colour Code for Fluorescent Lamps
The last three digits on many fluorescent lamps represents an international method of specifying a lamps colour rendering (Ra) and colour appearance
(colour temperature).
- The first digit is the colour rendering decade. Eg: 9 = 90 to 99), (8 = 80 to 89)
- The second digit is the colour appearance x100. Eg: 30 = 3000 degree K
Triphosphor
Standard fluorescent lamps (halophosphor) have only one type of phosphor powder coating the inside of the tube. The purpose of this coating is to convert the ultra violet light that is produced inside the lamp by electric energy, into visible light. With only one type of coating the light produced will not have very good colour rendering properties.
Triphosphor fluorescent lamps have 3 different phosphors coating the inside of the lamp. Each phosphor operates in its own colour band (red, green & blue) to give an improved combined colour output.
Such lamps produce about 10% more light output and can be made with better colour rendering ability and higher temperatures (colour appearance) than lamps with one phosphor.
Tri Color LED
A Tri Color LED is an LED that can light up into two different colors depending on where current flows to on the LED. A typical LED can only light up to one color. For example, a red LED can emit only a red light. A green LED can emit only a green light. However, with a tri color LED, two different colors can be lit with the same LED.

Tri color LEDs come in either 2 or 3 pins or terminals.
Below is the schematic for a 2 terminal tri color LED:
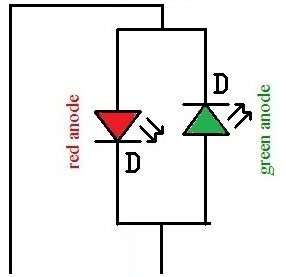
In the 2 terminal type, the LEDs are connected anode to cathode. Which LED lights depends on which terminal is made positive. If positive voltage is supplied to the green anode, the LED lights green. If positive voltage is supplied to the red anode, the LED lights red.
The other type is the 3 terminal tri color LED. Below is a schematic for it:
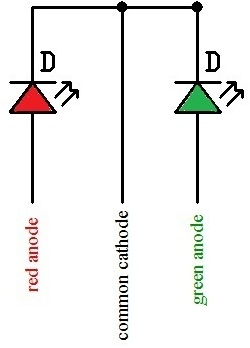
In the 3 terminal tri color LEDs, the LEDs have a common ground or cathode. A positive voltage applied to either of the other two wires is used to light the corresponding LED. In order to light the green LED, current from the circuit (a few milliamperes) must be made to flow into the green anode. In this setup, the circuit will light green light. In order to light the red LED, current from the circuit (a few milliamperes) must be made to flow into the red anode. While lighting one anode, the other anode should be kept open, meaning no current should go into it.
A tri color LED is useful as an indicator. It can indicate the mode in which an electronic device is operating. For example, on a digital camera, a tri color LED may glow red when it is in recording mode and green when it is in play mode.
Anyway, the most used lamp for shooting is daylight and bi-color.

Such as Portrait Shooting, Still Life Shooting, and Broadcasting.